Research in this section involves measurement, technique and instrument development and computation, with applications right across chemistry and with strong links to biosciences, chemical engineering, computer science, environmental science, materials science, medicine and physics.
We have particular expertise and interests in analytical science; astrochemistry; atmospheric chemistry; bio-nanotechnology; biophysical chemistry; catalysis; charge transfer and transport; clusters and nanoparticles; electrochemistry; environmental chemistry; fluorescence; magnetic resonance spectroscopy and imaging; optical sensors; reaction kinetics and dynamics; scanning probe microscopy; self-assembly; simulation and modelling; single molecule imaging; soft matter; solvation effects; spectroscopy; surface and interfacial chemistry; synchrotron-based characterisation; theoretical chemistry.
Research section leader

School of Chemistry
Chair in Cell Biology of Membrane Proteins
Department of Cardiovascular Sciences
- Email
- d.herten@bham.ac.uk
Areas of interest
Microscopy technique development, single molecule imaging, biophysical chemistry, molecular counting, fluorescent probe development
Representative publication: Live‐Cell Localization Microscopy with a Fluorogenic and Self‐Blinking Tetrazine Probe
P. Werther, K. Yserentant, F. Braun, N. Kaltwasser, C. Popp, M. Baalmann, D.-P. Herten and R. Wombacher
Angew. Chem. Int. Ed. (2020), 59, 804.
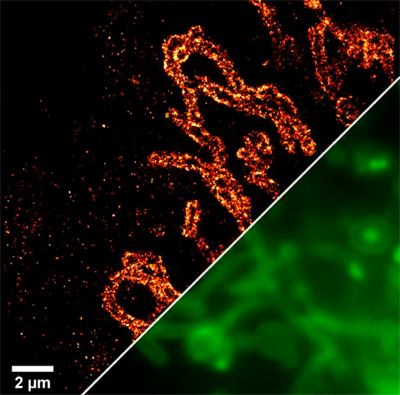
In this paper f‐HM‐SiR is presented as a small-molecule-based label with high functionalization allowing for excitation-independent super-resolution imaging with minimal phototoxicity in live-cell experiments. The probe is the first to combine fluorogenicity, photostability, high brightness, self-blinking, bio-orthogonal conjugation and far-red emission in a single label, making it highly favourable for single molecule localization microscopy in live cells. Using this probe greatly improved the imaging quality by reducing unspecific fluorescence (fluorogenicity) while allowing for long time imaging using low excitation powers (self-blinking).
Research section members
Areas of interest
Analytical chemistry; bio-nanotechnology; biophysical chemistry; catalysis; charge transfer and transport; clusters and nanoparticles; electrochemistry; scanning probe microscopy; self-assembly; single molecule imaging; soft matter; surface and interfacial chemistry
Representative publication: Unsupervised vector-based classification of single-molecule charge transport data
Mario Lemmer, Michael S. Inkpen, Katja Kornysheva, Nicholas J. Long & Tim Albrecht
Nature Communications 7 (2016) Article number: 12922
In this paper, we outline a new approach to analysing single-molecule charge transport and other data, namely by Multi-Parameter Vector Classification (MPVC). This allows for largely unsupervised classification of large datasets, essentially without making any a priori assumptions of what they look like.
Areas of interest:
Analytical chemistry; batteries; electrochemistry; magnetic resonance spectroscopy and imaging; simulation and modelling; soft matter
Representative publication: Operando visualisation of battery chemistry in a sodium-ion battery by 23Na magnetic resonance imaging
Joshua M. Bray, Claire L. Doswell, Galina E. Pavlovskaya, Lin Chen, Brij Kishore, Heather Au, Hande Alptekin, Emma Kendrick, Maria-Magdalena Titirici, Thomas Meersmann & Melanie M. Britton
Nature Communications 11 (2020) 2083
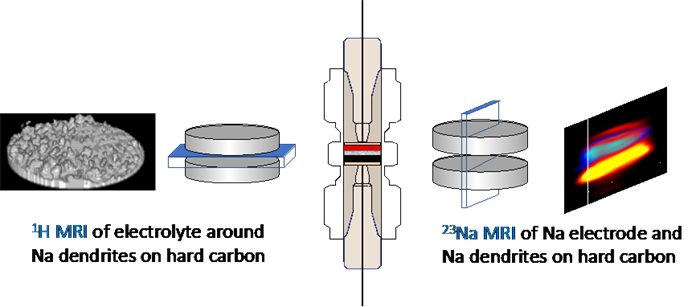
Sodium ion batteries (NIBs) offer significant cost and sustainability benefits over current lithium ion batteries, while maintaining high energy densities. Yet, there remain many challenges preventing their commercialisation, which is driving the development of improved SIB electrodes and electrolytes, as well as better understanding of the solid-electrolyte interphase and dendrite formation. Yet, the search for, and optimisation of, new sodium ion battery materials requires detailed measurement and characterisation, in operando, of each battery component, as well as the interactions and synergies between components and the causes of battery degradation. These detailed observations are currently lacking and inhibiting progress. This paper addresses this deficiency and reports the development of in operando 23Na magnetic resonance imaging (MRI) to enable, for the first time, direct visualisation of battery chemistry in a NIB, an exploration of the synergies between multiple components within the NIB and an assessment of the behaviour of a novel non-graphitic carbon SIB electrode material. This paper reveals new information about the environment and distribution of sodium within the NIB, which is correlated with the electrochemical behaviour of the cell. We observe, for the first time, the formation of sodium dendrites during galvanostatic plating in a sodium-metal cell and, more surprisingly, metallic sodium species on hard carbon upon first charge (formation) in a full-cell configuration.
Areas of interest
Active colloids; colloidal open crystals; colloidal self-assembly; computation and theory of soft materials; discotic liquid crystals; hierarchical self-assembly; multifunctional materials; photonic band gap materials; rheology of soft matter; topological soft matter
Representative publication: Leveraging Hierarchical Self-Assembly Pathways for Realizing Colloidal Photonic Crystals
Abhishek B. Rao, James Shaw, Andreas Neophytou, Daniel Morphew, Francesco Sciortino, Roy L. Johnston and Dwaipayan Chakrabarti
ACS Nano 14 (2020) 5348-5359

This computational study demonstrates a novel route to a much sough-after photonic crystal, exploiting hierarchical self-assembly pathways for designer colloidal particles via uniform clusters to promote crystallisation. The versatility of this bottom-up route to yield a variety of colloidal open crystals has been established in a series of recent studies.
Image credit: Wesley Flavell and Andreas Neophytou
Areas of interest
Functional molecules and materials, porous materials for challenging molecular separations; organic photocatalysts for chemical transformations; topology-driven design of covalent organic frameworks; optimization-driven design of experiments; explainable AI in chemistry; interactive data visualization
Representative publication: Combining machine learning and high-throughput experimentation to discover photocatalytically active organic molecules
Xiaobo Li, Phillip M. Maffettone, Yu Che, Tao Liu, Linjiang Chen and Andrew I. Cooper
Chem. Sci. (2021), 12, 10742–10754
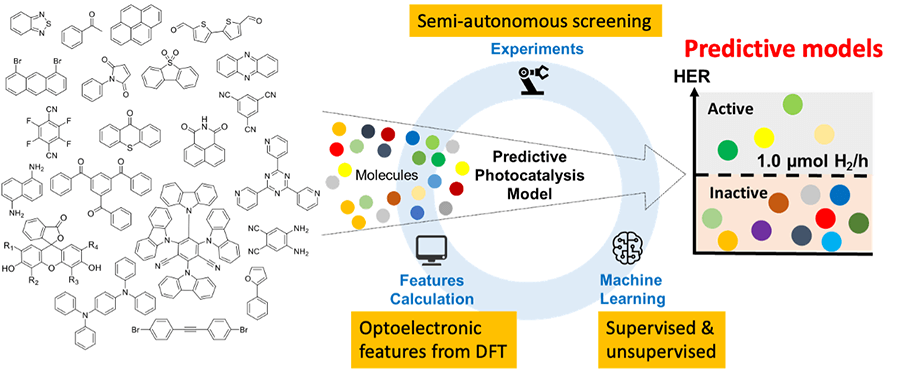
In this paper, we assembled the largest library of organic photocatalysts studied experimentally to date (as of April 2022) and tested all 668 of these molecules under identical experimental conditions for sacrificial photocatalytic hydrogen evolution. By applying unsupervised learning to atomic neighbour environments, we identified a correlation between molecular structure and photocatalytic activity that is human interpretable. We also used a suite of supervised classification algorithms, together with calculated molecular descriptors, to construct predictive models for hydrogen evolution rates, which allows in silico pre-evaluation of candidate molecular photocatalysts – effectively, an AI recommendation engine for selection of new catalysts in the future. This also revealed physical insights into the design rules for these molecular photocatalysts. This dataset of structures, properties, and calculated descriptors will be of interest to communities in both photocatalysis and in machine learning. With this in mind, we developed an interactive browser-based explorer for the molecular library that can be used by researchers with no experience in machine learning, Small-molecule photocatalysts. Virtual experiments using an adaptive machine learning model showed that the experimental cost of finding 50% of the active catalysts was decreased by a factor of two compared to random selection. This illustrates the potential of machine-learning-driven advisors to assist chemists, or autonomous robots, in the discovery of new photocatalysts.
Areas of interest
Electronic and structural properties of metal-organic interfaces; organic corrosion inhibitors; conjugated polymers; halogen bonds, 2D metal-organic networks, supramolecular self-assembly; surface science; surface and interfacial chemistry; scanning tunnelling microscopy (STM); X-ray photoelectron spectroscopy (XPS); electrospray deposition.
Representative publication: The Effect of Glycol Side Chains on the Assembly and Microstructure of Conjugated Polymers
Stefania Moro, Nicholas Siemons, Oscar Drury, Daniel A. Warr, Thomas A. Moriarty, Luís M. A. Perdigão, Drew Pearce, Maximilian Moser, Rawad K. Hallani, Joseph Parker, Iain McCulloch, Jarvist M. Frost, Jenny Nelson & Giovanni Costantini
ACS Nano 16 (2022) 21303–21314.
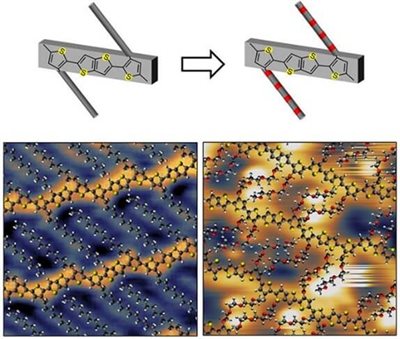
In this work, we determine how the microstructure of a high-performance conjugated polymer changes by modifying its side chains from alkyl to ethylene glycol. By using a combination of electrospray deposition and scanning tunnelling microscopy, we are able to acquire molecularly resolved images of the polymers. These reveal that the driving force for the polymer assembly in the tendency of the side chains to adopt the conformation of their free analogues, i.e., polyethylene and polyethylene glycol, for alkyl or ethylene glycol side chains, respectively.
Group web page
Ruchi Gupta Group
Areas of interest
Analytical science, optical sensors, microfluidics, micro-/nano-fabrication.
Representative publication: Method for Determining Average Iron Content of Ferritin by Measuring its Optical Dispersion
R. Gupta, N.A. Alamrani, G.M. Greenway, N. Pamme, N.J. Goddard,
Analytical Chemistry, 2019, 91, 7366. DOI: 10.1021/acs.analchem.9b01231.

We report a method to determine the average iron content of ferritin based on optical dispersion i.e., how strongly the refractive index changes with wavelength of light. The method is a significant advance over the state of the art method, ELISA used in clinics, because it allows determining the average content of ferritin in a single step. The average iron content of ferritin is an important marker for conditions such as injury, inflammation, and infection.
Areas of interest
Biophysical chemistry; catalysis; charge transfer and transport; clusters and nanoparticles; electrochemistry; environmental chemistry; self-assembly; surface and interfacial chemistry; synchrotron-based characterisation
Representative publication:Effect of Deuteration on Phase Behaviour of Supported Phospholipid Bilayers: A Spectroelectrochemical Study
E. Madrid and S.L. Horswell
Langmuir 31 (2015) 12544-12551
This paper is a study of electric field-induced structural changes observed in asymmetric lipid bilayers, using in situ electrochemical PM-IRRAS. A key potential tool for discriminating between different components of lipid layers is selective deuteration, which is sometimes used in infrared spectroscopy and is standard in neutron studies. This IR study employs selective deuteration to study each half of the bilayer separately. Its significance is that we found that deuteration can alter properties of the ensemble of molecules. Our results mean that care has to be taken when utilising isotopic substitution because even if isotherms of monolayers seem to indicate similar properties in the condensed phases, differences in behaviour of bilayers from which they are formed are still possibilities that should be considered.
Group web page
Lehman Group
Areas of interest
Astrochemistry, atmospheric chemistry, high resolution infrared spectroscopy, photodissociation dynamics, gas phase chemical reaction kinetics, frequency comb laser spectrometers
Representative publication: Infrared frequency comb spectroscopy of CH2I2: Influence of hot bands and pressure broadening on the ν1 and ν6 fundamental transitions
F. C. Roberts, J. H. Lehman, Journal of Chemical Physics, 156, 114301 (2022); https://doi.org/10.1063/5.0081836
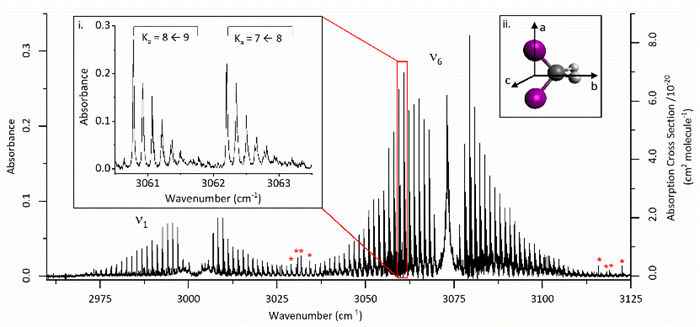
This is the second paper published using our newly built frequency comb spectrometer, showcasing several superior attributes of using frequency comb spectroscopy for obtaining high resolution gas phase spectra over other techniques. From the abstract: “We report the vibrational absorption spectrum of diiodomethane, CH2I2, from 2960 to 3125 cm−1. The rovibrational spectra of two fundamental transitions, ν6 and ν1, were recorded and analysed. We suggest that a significant contribution to the observed congested spectra is due to the population in excited vibrational states of the low energy ν4 I–C–I bend, resulting in transitions 6104nn and 1104nn, where the integer n is the initial vibrational level v = 1–5. PGOPHER was used to fit the experimental spectrum, allowing for rotational constants and other spectral information to be reported. Further implications for other dihaloalkane infrared spectra are discussed.
Areas of interest
DNA methyltransferases, epigenetics; fluorescence; biophysical chemistry and single molecule imaging
Representative publication: Site-selective and Re-writable Labeling of DNA through Enzymatic, Reversible and Click Chemistries
Andrew A. Wilkinson, Elodie Jagu, Krystian Ubych, Steven Coulthard, Ashleigh E. Rushton, Jack Kennefick, Qiang Su, Robert K. Neely and Paco Fernandez-Trillo, (2020)
ACS Central Science, 6, 4, 525-534

The paper, a collaboration with Dr Paco Fernandez-Trillo, describes a novel chemistry for the reversible modification of DNA at specific sites of interest. We show that DNA can, for example, be labelled sequentially with two different coloured fluorophores. In the lab we are further developing the method to tag, capture and purify DNA for analysis using DNA sequencing. We hope this approach will find application in the early-stage diagnosis of many diseases in the future.
Softley Group: "Chemical dynamics at ultracold temperatures"
Overview
We are developing new machine-learning approaches to the real-time analysis of fluorescence-images of Coulomb crystals – assemblies of 10s or 100s of laser-cooled ions in a Paul Trap that form ellipsoidal-shaped structures under the cooling/trapping conditions. We use these crystals as a medium for studying the kinetics and dynamics of chemical reactions at ultracold temperatures, following reactions by observing changes in the Coulomb crystal structure.
Categories: imaging, deep learning, transfer learning
Contact
Enquiries about specific aspects of their research areas should be addressed to individual research group leaders. For more general enquiries about working with the I2SUnit, please contact the Head of the Section.