Developmental Neuroscience Group
Overview
The Tuxworth group studies the fundamental cellular processes required for the development of the nervous system and for maintenance of neural function. The group has a particular focus on lysosomal biology and the role lysosomes play in neural function.
Our Research
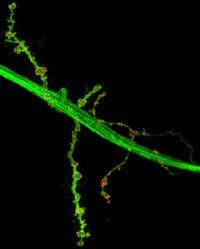 The Tuxworth group’s interest in lysosomal function in the nervous system has developed from study of the lysosomal storage disorders – a group of rare inherited syndromes that display lysosomal dysfunction. In many of these disorders neural pathology develops early in life, indicating that the nervous system is particularly sensitive to lysosomal dysfunction. In normal ageing and in late-onset neurodegenerative conditions such as Alzheimer’s and Parkinson’s disease, reduction in lysosomal function is also likely to contribute to the decline in neural activity.
The Tuxworth group’s interest in lysosomal function in the nervous system has developed from study of the lysosomal storage disorders – a group of rare inherited syndromes that display lysosomal dysfunction. In many of these disorders neural pathology develops early in life, indicating that the nervous system is particularly sensitive to lysosomal dysfunction. In normal ageing and in late-onset neurodegenerative conditions such as Alzheimer’s and Parkinson’s disease, reduction in lysosomal function is also likely to contribute to the decline in neural activity.
The main focus of our research currently is on Batten disease (or neuronal ceroid lipofuscinsoses), a group of fatal inherited forms of childhood-onset neurodegenerative disease. Since pathology manifests very early in life in Batten disease patients and is almost exclusively restricted to the nervous system, we are exploring the possibility of a developmental component to the disease. We predominantly use simple fruitfly (Drosophila) models of Batten disease where changes in the neurodevelopment can be observed and quantified easily using microscopy and cell biology techniques.
A second strand of research started recently within our group looks at neurodegeneration at the other end of the life course. With colleagues in the Institute for Cancer and Genomics we are exploring the importance of DNA damage in the development of neural pathology in late-onset forms of neurodegeneration, including Alzheimer’s disease, Frontotemporal dementia, Huntingdon’s disease and amyotrophic sclerosis.
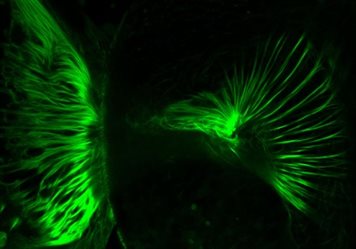
- The importance of lysosomal function for neural development and synaptic function
(Dr Niki Anthoney, Mathew Taylor, Alamin Mohammed)
This project examines the consequences for neural development of loss-of-function mutations in NCL genes, including CLN3 and CLN7. We are investigating the signalling pathways that drive neural development and asking how they impinge on NCL gene function.
- In situ proximity labelling of lysosomal membrane proteins
(Kyle Connolly)
This project uses a proteomics approach to identify the components of protein complexes in the lysosomal membrane. We ask how these components change as cells are exposed to varying environmental conditions and help explain changes the see in neural development in NCL models.
- Structural biology of the CLN3 protein
(Dr Niki Anthoney)
This project aims to express, purify and solve the three dimensional structure of the CLN3 protein. CLN3 mutations cause Juvenile NCL (Batten disease), the most common form of NCL. The protein is a transmembrane protein of unknown function.
- The importance of the DNA damage response in neuropathology
(Mathew Taylor)
We examining the consequences for neural health and function of activating the DNA damage response. We are using Drosophila models of late-onset neurodegenerative conditions and genetic tools to dissect the damage response pathway and its effect on pathology.
Faller KM, Gutierrez-Quintana R, Mohammed A, Rahim AA, Tuxworth RI, Wager K and Bond M (2015) The Neuronal Ceroid Lipofuscinoses: opportunities from model systems. Biochim Biophys Acta 1852(10 Pt B):2267-78
Povellato G*, Tuxworth RI*, Hanger DP and Tear G (2013) Modification of the Drosophila model of in vivo Tau toxicity reveals protective phosphorylation by GSK3b. Biol Open 3(1):1-11
*Joint first authorship
Tuxworth RI, Chen H, Vivancos V, Carvajal N, Huang X and Tear G (2011) The Batten disease gene CLN3 is required for the response to oxidative stress. Hum Mol Genet 20(10):2037-47
Tuxworth RI, Vivancos V, O’Hare MB and Tear G (2009) Interactions between the juvenile Batten disease gene, CLN3, and the Notch and JNK signalling pathways. Hum Mol Genet 18(4):667-78
Principal Investigator
Dr Richard Tuxworth - Lecturer in Molecular Genetics, Institute of Cancer and Genomic Sciences
Internal Collaborators
Dr Zubair Ahmed - Senior Lecturer in Neuroscience, Institute of Inflammation out of Ageing
Dr Timothy Knowles - Lecturer in Biophysics, School of Biosciences
Postdoctoral Researchers
Dr Niki Anthoney
PhD Students
Alamin Mohammed
Kyle Connolly
Research technician
Mathew Taylor
External Collaborators:
Professor Guy Tear - King's College London